With the opioid crisis in the United States, we have seen increasing rates of opioid use across multiple sectors of our population, including women of reproductive-age. The risks of opioid use are significant, especially during pregnancy, when opioid use is associated with higher risk of pregnancy loss, adverse pregnancy outcomes, and neonatal abstinence syndrome.
In 2017, approximately 8.2 per 1000 deliveries were affected by opioid use disorder in this country, with even higher rates observed in women who were publicly insured, with an estimated 14.6 per 1000 deliveries affected. Standard care for treating opioid use disorder during pregnancy is opioid agonist therapy with either buprenorphine or methadone.
Medication treatment of opioid use disorder during pregnancy has unequivocal benefits and is associated with improved adherence to prenatal care, lower incidence of preterm birth, reduced recurrence of opioid use, and decreased risk of opioid overdose and death from opioid overdose. Traditionally methadone has been used for the treatment of OUD during pregnancy; more recently buprenorphine has become a first-line treatment.
However, there are important differences between the two options. Methadone is a full agonist with high activity at mu opioid receptors, whereas buprenorphine is a high-affinity partial agonist with low activity at mu opioid receptors. While methadone must be administered during daily in-person visits to federally regulated opioid treatment programs; buprenorphine may be prescribed by approved providers and self-administered by patients. A recent study looks at the differences between these two treatment options in terms of neonatal and maternal outcomes.
In this study, Suarez and colleagues used nationwide Medicaid data to identify pregnancies from 2000 through 2018. They evaluated maternal and neonatal outcomes among patients who received buprenorphine monotherapy or buprenorphine-naloxone combination therapy versus methadone. Neonatal abstinence syndrome was assessed in relation to treatment administered in the 30 days preceding delivery.
The study included 2.5 million pregnancies resulting in a live birth. In early pregnancy, 10,704 patients were exposed to buprenorphine and 4,387 to methadone; in late pregnancy (week 20 or later), 11,272 and 5,056 were exposed, respectively. The majority of pregnant patients who were exposed to medication-assisted treatment in late pregnancy received treatment within 30 days of delivery.
Fewer Adverse Events in Buprenorphine-Exposed Pregnancies
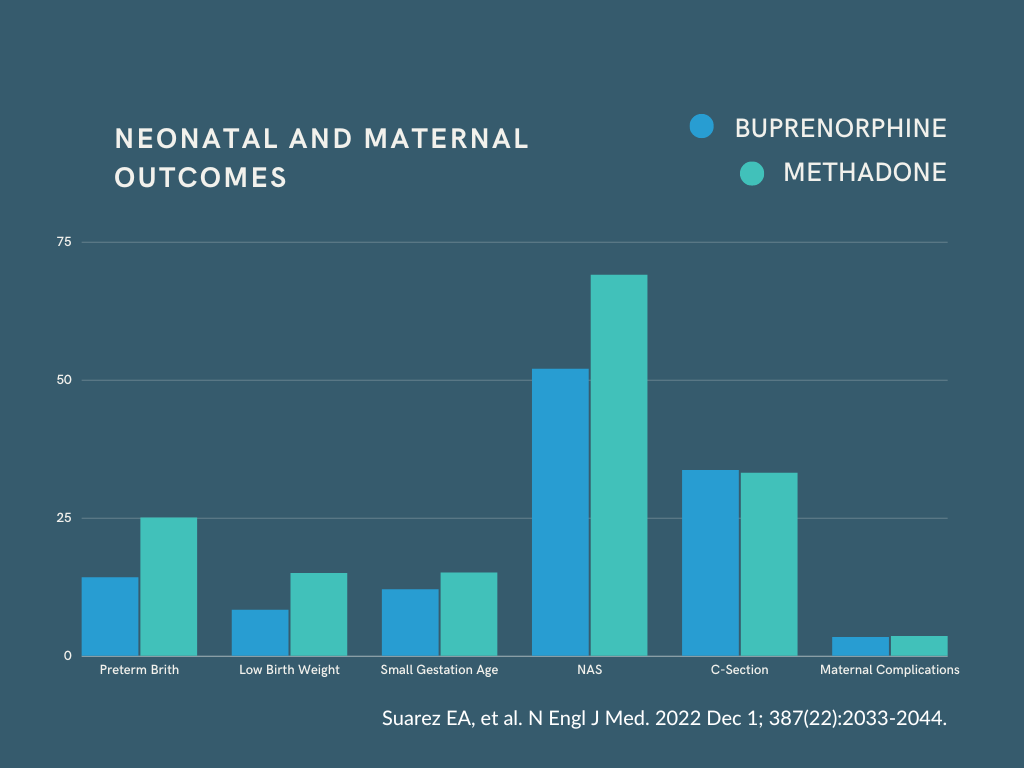
Neonatal abstinence syndrome occurred in 52.0% of the infants who were exposed to buprenorphine in the 30 days before delivery as compared with 69.2% of those exposed to methadone (adjusted relative risk, 0.73; 95% CI, 0.71 to 0.75).
Preterm birth occurred in 14.4% of infants exposed to buprenorphine in early pregnancy and in 24.9% of those exposed to methadone (adjusted relative risk, 0.58; 95% CI, 0.53 to 0.62)
Small size for gestational age in 12.1% and 15.3%, respectively (adjusted relative risk, 0.72; 95% CI, 0.66 to 0.80); and
Low birth weight in 8.3% and 14.9% (aRR, 0.56; 95% CI, 0.50 to 0.63).
Rates of delivery by cesarean section were similar in the two groups: 33.6% in buprenorphine- vs. 33.1% in methadone-exposed pregnancies (aRR, 1.02; 95% CI, 0.97 to 1.08).
Rates of severe maternal complications were similar in the two groups: 3.3% and 3.5%, respectively (aRR 0.91; 95% CI, 0.74 to 1.13).
Clinical Implications
The current study — the largest to date — observes a lower risk for adverse neonatal outcomes with the use of buprenorphine compared to methadone used during pregnancy in a large cohort of individuals with opioid use disorder. However, the risk of adverse maternal outcomes was similar among persons who received buprenorphine and those who received methadone. While methadone used to be used more commonly, the results of this study indicate that buprenorphine should be considered as a first-line treatment for women who are pregnant or of childbearing age. While buprenorphine is associated with fewer adverse events, the authors remind us that any treatment for opioid use disorder in pregnancy is better than no treatment.
In an accompanying editorial, Elizabeth Krans, MD, of the University of Pittsburgh, discusses some of the challenges in providing care to pregnant patients with OUD, noting that the decision to use buprenorphine or methadone must reflect patient preferences, previous treatment, and medication availability. She also notes that as there has been an increase in the use of high-potency synthetic opioids like fentanyl, treatment with buprenorphine is more challenging. Because buprenorphine is a partial opioid receptor agonist, it may not completely mitigate opioid cravings and could cause withdrawal in patients who use fentanyl.
Ideally decisions regarding the selection of optimal treatment of opioid use disorder should take place before pregnancy. However, approximately 80% of pregnancies in women with OUD are unplanned. In addition, many women initiate treatment with medication for OUD after conception. Whatever choices are made, we have to support our patients who are pregnant or planning to conceive, reminding them that either treatment is better than no treatment.
Ruta Nonacs, MD PhD
Suarez EA, Huybrechts KF, Straub L, Hernández-Díaz S, Jones HE, Connery HS, Davis JM, Gray KJ, Lester B, Terplan M, Mogun H, Bateman BT. Buprenorphine versus Methadone for Opioid Use Disorder in Pregnancy. N Engl J Med. 2022 Dec 1; 387(22):2033-2044.